Is it possible to transmute lead into gold? Scientific American explains that “it is indeed possible—all you need is a particle accelerator, a vast supply of energy and an extremely low expectation of how much gold you will end up with.”
For scientists and teachers who don’t have all that, there’s the golden rain experiment. In this video, Exploratorium Senior Scientist Julie Yu mixes lead nitrate Pb(NO3)2 (toxic to your health) with potassium iodide KI to create gold-colored lead iodide PbI2 (also toxic to your health). The short video provides an excellent demonstration of this dangerous experiment.

What’s going on in this ionic bonding demo? A golden rain summary from Compound Chemistry:
This mixing leads to a double displacement reaction, essentially resulting in the metals ‘swapping’ their places in the two compounds, producing lead (II) iodide, and potassium nitrate. It also produces a rapid colour change, as the lead iodide is very insoluble in water at room temperature. When the solutions are added together, they immediately produce a bright yellow precipitate of lead iodide…
These hexagonal crystals take some time to meander gently to the bottom of the flask, giving the reaction mixture a shimmering, glittering effect commonly referred to as a ‘golden rain’.
The effect can last for up to an hour as the crystals fall out of the solution, so it’s a great experiment to instill an awe of chemistry!
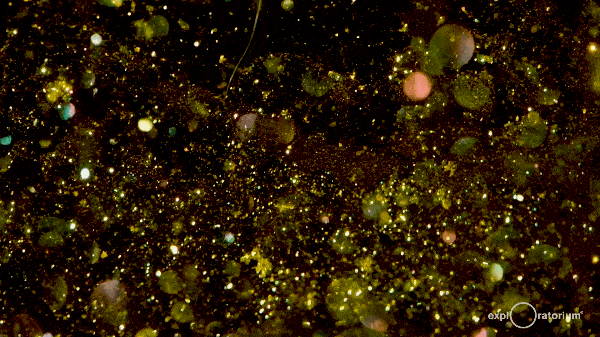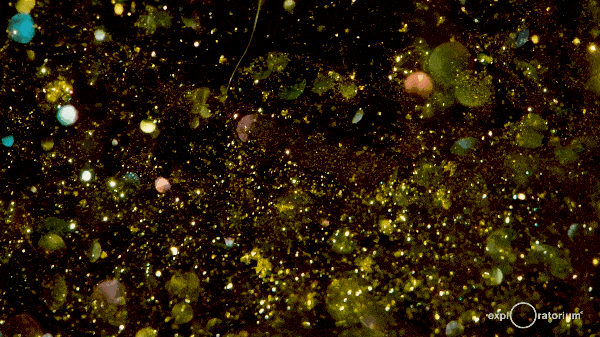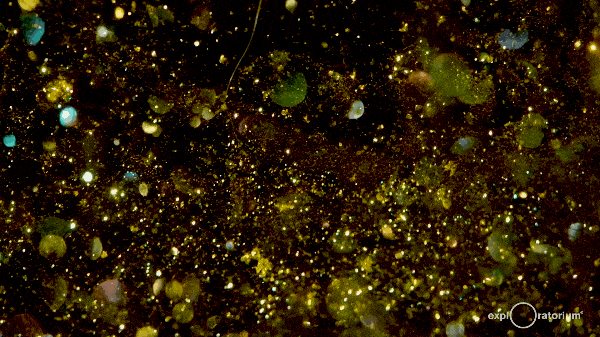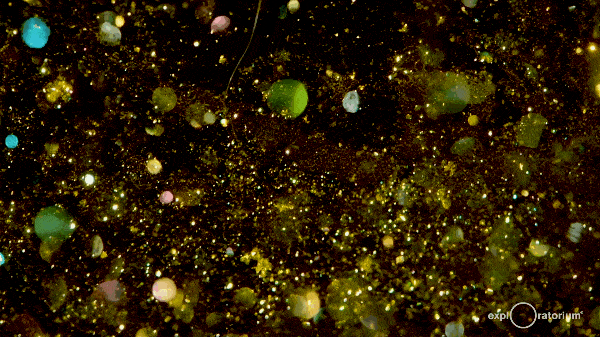
There’s more information at both Education in Chemistry and The Home Scientist.
Plus, watch more chemistry videos and more demonstration videos on this site, including Making ice cream with Liquid Nitrogen and Dr. Kate Biberdorf and using a 9-volt battery to break water into its elemental components.
And from The Ring of Truth: Molten gold transforms into gold leaf.
Curated, kid-friendly, independently-published. Support this mission by becoming a sustaining member today.

